Get Healthy!
- Posted April 2, 2026
New Pill Could Change Plaque Psoriasis Treatment
Folks with severe plaque psoriasis often have to choose between convenient pills that don’t work very well or highly effective injections that come with the hassle of needles.
That trade-off may soon change. New clinical trial data suggests a once-daily pill called zasocitinib may provide the clear skin once only expected from shots.
The medicine is in the final stages of study and is not yet approved by the U.S. Food and Drug Administration (FDA).
Takeda Pharmaceuticals recently shared results from two late-stage clinical trials involving nearly 1,800 adults with moderate-to-severe plaque psoriasis across 21 countries.
The findings -- presented at a meeting of the American Academy of Dermatology in Denver last month -- showed that about 70% of participants taking the pill reached the goal of having clear or almost clear skin within 16 weeks.
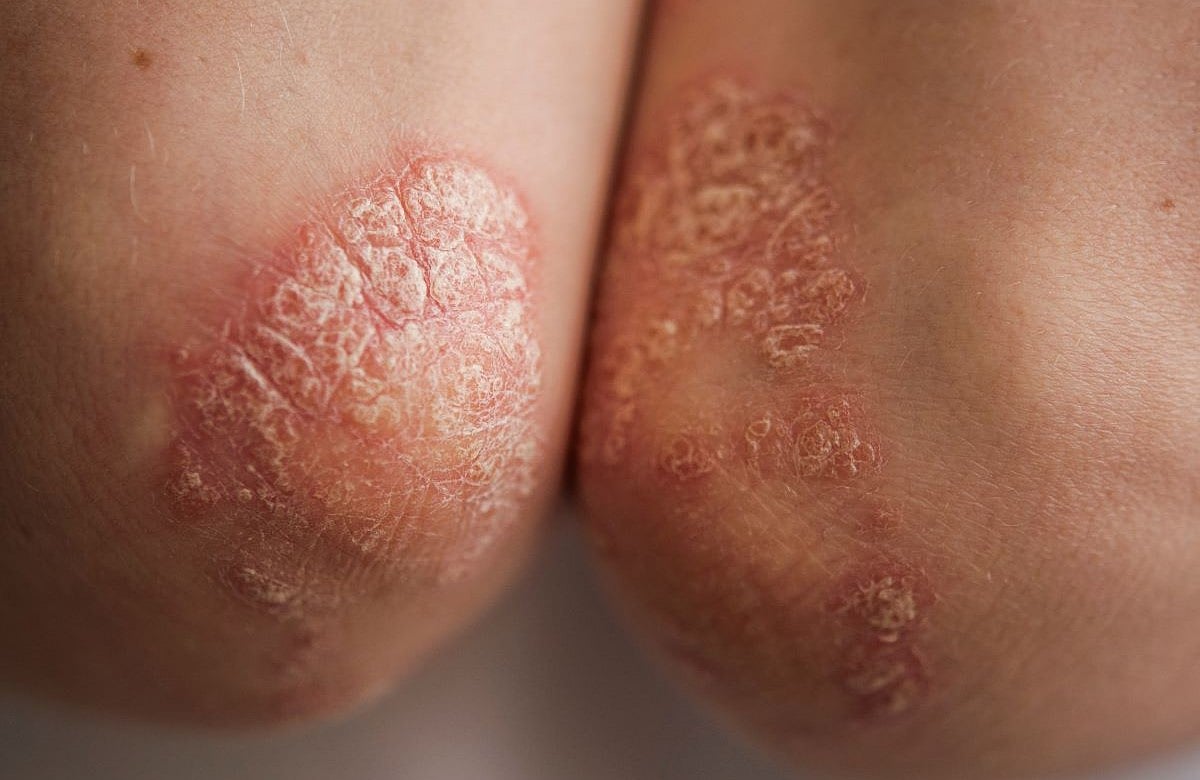
Plaque psoriasis makes skin cells grow too fast, forming thick, scaly raised red patches with silvery scales. It causes itching and burning feelings on the skin during flare-ups.
The studies compared zasocitinib to a placebo and to apremilast, a widely available pill for psoriasis.
About 30% of patients on apremilast saw significant skin improvement, compared to more than double that amount with zasocitinib.
Researchers reported that zasocitinib worked with surprising speed.
Many patients noticed their skin starting to clear as early as four weeks into the treatment.
The results were lasting: Of the patients who had clear skin at the nine-month mark, more than 90% still maintained those results after more than a year of daily use.
“Our goal in psoriasis treatment is clear or almost clear skin, and previously this has been achieved primarily with injectable therapies,” said lead researcher Dr. Melinda Gooderham, a dermatologist in Ontario, Canada.
She added that these results show it is possible for a daily pill to deliver rapid, lasting relief.
No new or unexpected safety concerns were discovered during the trials. The most common side effects reported were generally mild, such as upper respiratory infections like a common cold.
About 6.5% of patients taking the drug also experienced acne, which is a known side effect of this specific class of medication, called TYK2 inhibitors.
“Our Phase 3 results demonstrate that highly selective TYK2 inhibition can offer many people with moderate-to-severe plaque psoriasis the potential for clear or nearly clear skin,” said Dr. Chinweike Ukomadu, a senior vice president at Takeda and head of the company’s gastrointestinal and inflammation areas.
Takeda plans to submit the drug to the FDA for approval within the next year.
Findings from medical conferences are considered preliminary until published in a peer-reviewed journal.
More information
The National Institute of Arthritis and Musculoskeletal and Skin Diseases has more on psoriasis.
SOURCE: Takeda Pharmaceutical Company Limited, news release, March 28, 2026




